Clinical Evidence supporting the NowMi Treatment
The NowMi treatment was tested in a clinical study performed by a third party GLP certified laboratory. The clinical study was intended to evaluate the safety and efficacy of the NowMi PRO treatment for improving skin texture, skin smoothens and for immediate and long-term overall skin appearance improvement when used in a controlled panel test.
The treatment protocol included:
Daily cleansing: six weeks of daily facial cleansing at night with the NowMi PRO sonic device and the NowMi facial cleansing gel.
Weekly treatments: six facial rejuvenation treatments carried out once a week with NowMi PRO sonic device that uses the vitamin C effervescent tablet and the NowMi lubricant gel.
Results:
Statistically significant results (p<0.05) obtained. After 3 weeks of treatments, facial skin smoothness increased by 15.1%, and after 6 weeks smoothness increased by 25.9%. No adverse effect were recorded and the safety and efficacy of the NowMi PRO treatment was proven.
The NowMi PRO Kit
The NowMi Pro has two functions – the NowMi Pro Daily is designed to keep the skin clean and moisturized on a daily basis, and the NowMi Pro Weekly provides a ‘spa-like’ deep facial that slows down the signs of aging, revitalizes the skin and protects it against extrinsic factors such as smoke and pollution.

Clinical Study Methodology
The methodological approach used to evaluate whether or not the product tested has any effect on facial skin rejuvenation included: Objective (instrumental) measurements for capturing and analyzing skin texture.
Following the application of a product on human subjects (see Colipa Guidelines Efficacy 2008), the instrumental tests are performed with instruments that can precisely measure given parameters, according to a defined principle. Measurements are made under the control of a suitably qualified professional and use precise criteria (Colipa Guidelines Efficacy 2008).
To capture and evaluate skin’s texture, Canlfield’s VISIA system was used. The VISIA system consists of the VISIA imaging booth and VISIA software and can capture and store facial images using standard lighting, cross-polarized flash, and UV flash. After capturing images, the software automatically isolates or “masks” specific areas of the face and then performs an extensive analysis of these areas to evaluate the skin features.
The study was carried out on a panel of 10 women, 8 with normal skin and 2 with sensitive skin. The women were aged 32.8±4.4 years.
The table below depicts the sex, age and skin type of the subjects participated in the study.
| Subject No. | Skin type | Sex | Age |
| 1 | Normal skin | Female | 30 |
| 2 | Normal skin | Female | 36 |
| 3 | Normal skin | Female | 30 |
| 4 | Normal skin | Female | 26 |
| 5 | Normal skin | Female | 26 |
| 6 | Normal skin | Female | 39 |
| 7 | Normal skin | Female | 34 |
| 8 | Normal skin | Female | 34 |
| 9 | Sensitive skin | Female | 38 |
| 10 | Sensitive skin | Female | 35 |
Results of objective measurements of skin texture
Texture is primarily an analysis of skin smoothness. It is affected by wrinkles, pore size and general roughness of the skin. The results of skin texture as measured by the VISIA system are presented in the table below for each volunteer.
| After 6th treatment | After 3rd treatment | Baseline | Volunteer No. |
| 287 | 358 | 438 | 1 |
| 113 | 74 | 113 | 2 |
| 77 | 98 | 80 | 3 |
| 447 | 531 | 630 | 4 |
| 736 | 701 | 812 | 5 |
| 285 | 294 | 354 | 6 |
| 222 | 265 | 292 | 7 |
| 126 | 185 | 168 | 8 |
| 281 | 337 | 553 | 9 |
| 257 | 401 | 383 | 10 |
| 283.10 | 324.40 | 382.30 | Average |
| 192.21 | 191.34 | 235.00 | St.dev. |
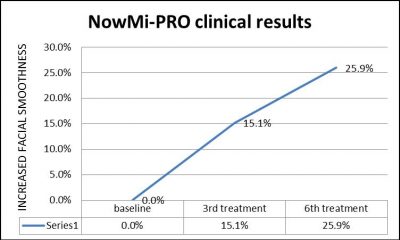
Conclusions:
- 70% of subjects presented increase in facial skin smoothness after 3 weekly treatments compared to baseline.
- 90% of subjects presented increase in facial skin smoothness after 6 weekly treatments compared to baseline.
- A statistically significant improvement of facial skin smoothness was measured both after 3 weekly treatments and at the end of the study compared to the baseline.
NowMi Pro is now on sale with Special Offer: Buy now a NowMi Pro Kit from the inventor of OxyGeneo and receive Free of Charge a NowMi refill kit (valued at $99) that will give you 3 extra months vitamin C oxygen facial treatments at home.